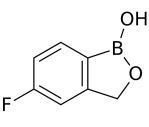
 The antifungal drug tavaborole was approved by the FDA a few weeks ago. One thing that is unusual about the chemical structure of tavaborole is the boron atom that it contains. Boron is not a common element in pharmaceutical drugs. The cancer drug bortezomib (Velcade) comes to mind, but there aren’t many. The antibiotic compound boromycin contains boron too, but it’s not a marketed drug.
The antifungal drug tavaborole was approved by the FDA a few weeks ago. One thing that is unusual about the chemical structure of tavaborole is the boron atom that it contains. Boron is not a common element in pharmaceutical drugs. The cancer drug bortezomib (Velcade) comes to mind, but there aren’t many. The antibiotic compound boromycin contains boron too, but it’s not a marketed drug.
So what’s wrong with boron that prevents its widespread use in pharmaceuticals? As far as I know, and also according to a review in EMBO Reports, there is nothing inherently bad about boron in terms of it being used in medicines. It seems to be that medicinal chemists simply don’t try to use it often.
The company that developed tavaborole, Anacor Pharmaceuticals, is trying to take advantage of this omission. Their whole pipeline of drug candidates are relatively simple organoboron compounds. You can carve out some novel chemical space in terms of intellectual property that way.
I have encountered a similar situation in my career, except instead of an underutilized element, it involved an underutilized functional group. In developing the anticonvulsants JNJ-26990990 and JNJ-26489112, we were able to patent some very simple* compounds because they contained the relatively uncommon sulfamide functional group. We encountered no inherent problems with druggability associated with the sulfamide groups, and noticed that it showed up relatively infrequently in the patent literature, so we came to the conclusion that it was an underappreciated and underutilized functional group in medicinal chemistry.
*Readers in the drug discovery field can imagine how pleased our scale-up group was to learn that our first clinical candidate could be prepared in one step from commercially available materials.